Hollow cathode lamp faas8/1/2023 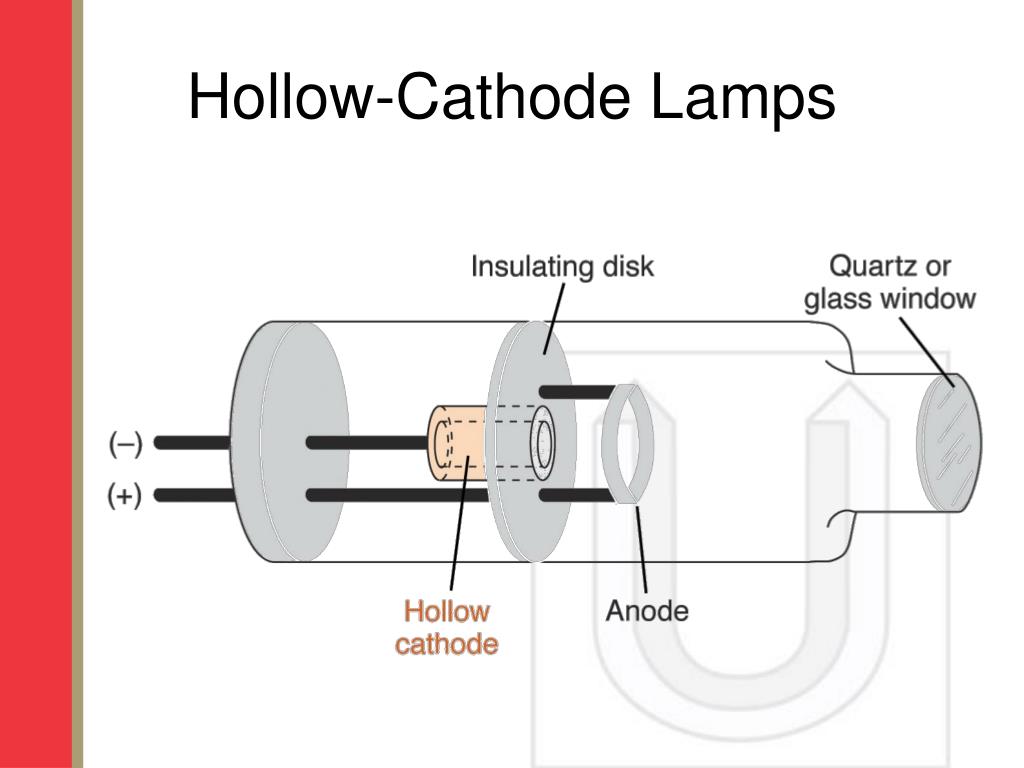
Therefore, the use of a continuum light source in AAS would obliterate the analytical specificity afforded by the technique: anything present in the sample that absorbs light in the region of overlap of (a) the emission wavelength range of the light source and (b) the wavelength range of sensitivity of the detector would be recorded in a given measurement. This is the cup-shaped metal electrode in the centre of the image. Information on typical results of a single element Atomic Absorption cathode lamp, based on the standard operating conditions for each lamp. As the name implies, the key component is a hollow cathode made or, or containing, the element to be analysed. Atomizing techniques - flame atomic absorption spectroscopy (FAAS)FAAS is. Hollow-cathode lamps contain a cathode of the analyte element and an anode, and are filled with a noble gas. The hollow cathode lamp (HCL) achieves this by using a high-energy discharge to stimulate emission from the element of interest. In AAS, any light that is passed through the sample has the potential to be absorbed by anything present in the sample. produced by a hollow-cathode lamp, and undergo electronic transitions from the.Hollow cathode lamps emit light that is specific to the element. While the "fingerprint" wavelengths of each element are highly specific, they often sit at wavelengths close to those of other elements. Flame atomic absorption spectrometry (FAAS) is a globally recognized analytical.Most detectors used in AAS / OES are broad-spectrum: they report just about any light that strikes them within their respective ranges of sensitivity, which usually span several hundred nanometers. Most instruments today use a hollow cathode lamp that is specific to each element being analyzed to emit a very narrow bandwidth of UV or visible radiation into.These lamps are encased in a cylinder made out of glass walls and a quartz end cap. A schematic of a Calcium HCL is shown in Animation 2.1 below. One of the strengths of atomic absorption and other ionizing elemental analysis techniques using optical detection ( ICP-OES is the main other one I know of) is that the optical absorption and emission properties at particular wavelengths are highly element-specific.ĭespite this, the reasons for desiring monochromatic, element-specific AAS light sources are as follows: The most common line source radiation generator used in AAS is the hollow cathode lamp (HCL).
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
